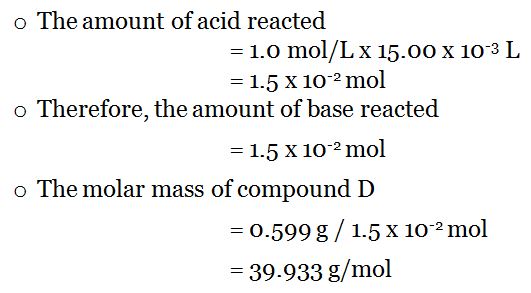
Learn more about acids in water, as well as other acids and bases.įind concentrated HCl density, concentrated HCl molarity, and more below! If you need smaller quantities, we have 30 ml bottles of hydrochloric acid for sale. 0025 moles HCl Now you work backwards with the concentrated solution. This chemical is used to adjust the pH of swimming pools to make them suitable for use and to disinfect the water it is often called "muriatic acid" in this context. First determine the amount of moles in the final 10 mL 0.25 M HCl. The mass of the earths atmosphere is estimated as 5.14 x 1015 t (1 t 1000 kg). 
The driving factor for solution formation is a) entropy b) enthalpy c) temperature d) molar mass of the. The molarity of HCl is 11.65 M find the number of moles in 0.70 mL. DETERMINATION OF THE MOLAR MASS OF A METAL (1m)Objectives (2m)To standardize the hydrochloric acid solution.To determine the molar mass of an alkaline earth. The equivalent weight of hydrochloric acid 36. See also our theoretical yield calculator for chemical reactions (probably your next stop to finish the problem set). 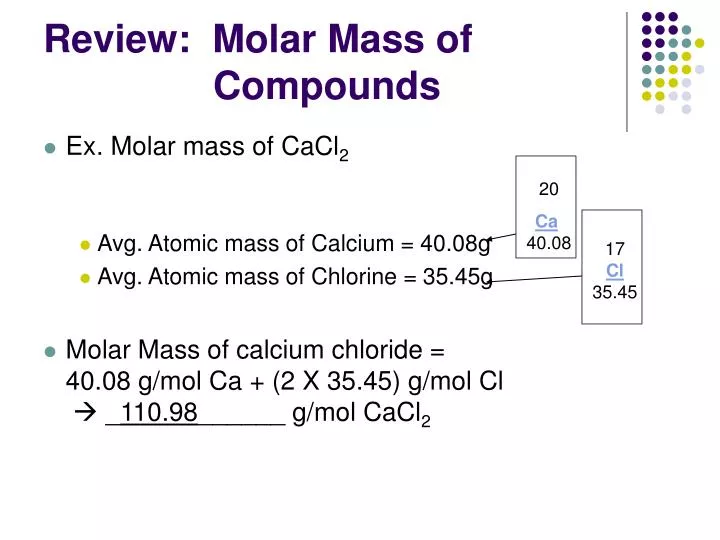
Concentrated HCl is a highly corrosive acid with many industrial uses. Access the answers to hundreds of Molar mass questions that are explained in a way thats easy for you to understand. The molar mass of HCl (hydrochloric acid) is: 36.458 grams/mol. One mole of a compound contains Avogadros number (6.022 x 1023) of molecules (molecular compound) or formula units (ionic compound). Hydrochloric acid is a colorless solution of hydrogen chloride (HCl) in water. Tris-hcl hydrochloride C4H13Cl2NO4562 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological. Hydrochloric Acid 36 -40 231-595-7 HCl 36 If the mass of the. This concentrated HCl solution is tech grade. DENSITY EXPANSION (DEX) APPROXIMATION FOR MIXTURE RDFs Molar mass of KOH 56. This 100 ml bottle of hydrochloric acid is a concentrated 12 Molar 32-36% solution. HCl is a colorless gas at room temperature.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
